PhD-Level Overview of Detoxination
Introduction
This review synthesizes the physiological basis, clinical applications, safety governance, and published human outcomes related to the Detoxination® protocol (Hubbard-method lineage). Citations are consolidated as numbered endnotes for clarity and web readability.
1. Historical Context & Lineage
2. Evidence Hierarchy & Methods
3. Physiology & Niacin-Driven Rebound Lipolysis
4. Protocol Application & Parameters
5. Human Outcomes with Hubbard-Method Programs
6. Safety, Contraindications & Governance
7. Comparative Approaches & Confounders
8. Limitations & Research Gaps
Endnotes
1. Historical Context & Lineage
Detoxination® grew from clinical work targeting lipophilic xenobiotic burden—including, but not limited to, persistent organic pollutants (POPs such as PCBs/dioxins), certain legacy and modern pesticides, selected solvents, and some pharmaceuticals—with a structured sequence of immediate-release niacin, timed physical activity, and sauna-mediated thermoregulatory sweating, complemented by hydration, electrolytes, micronutrients, and binders. Program lineages include addiction rehabilitation, occupational and disaster-exposure cohorts (e.g., law-enforcement and rescue workers) and later pilots in chronic, exposure-linked syndromes.[1,2,3,4]
2. Evidence Hierarchy & Methods
This review distinguishes between (a) foundational physiology, (b) observational human data, and (c) interventional trials. Clinical claims are labeled by evidence tier and limited to what is directly reported. Where inferential links are made (e.g., between free-fatty-acid surges and xenobiotic co-mobilization), this is stated explicitly.[5,6]
3. Physiology & Niacin-Driven Rebound Lipolysis
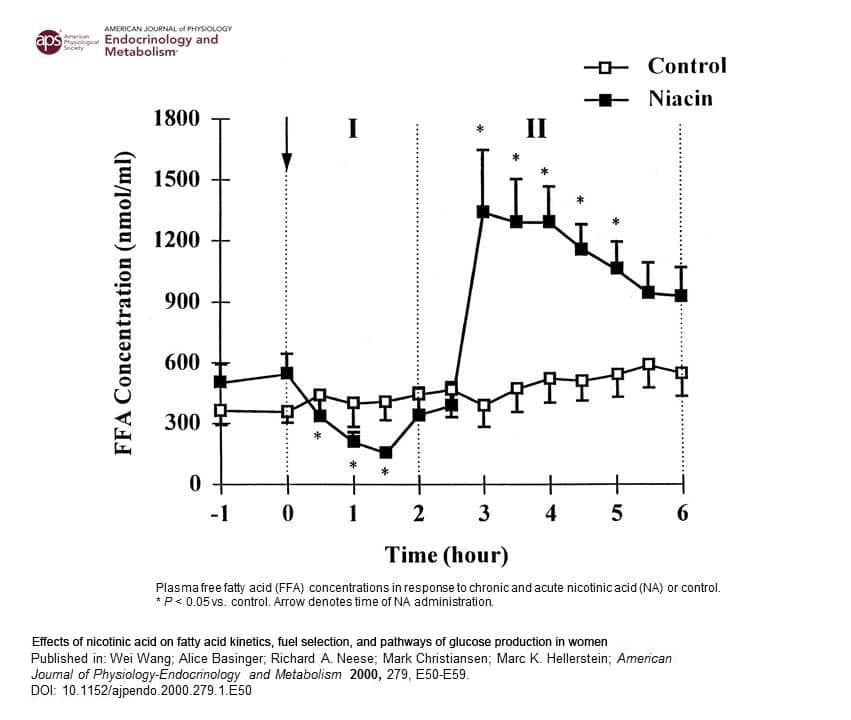
Mechanism. Immediate-release nicotinic acid transiently suppresses adipose lipolysis via GPR109A (HCA2), followed by a compensatory “rebound” increase in lipolysis and circulating free fatty acids (FFA). In human kinetics, a pronounced FFA elevation emerges several hours post-dose and gradually returns toward baseline thereafter; this timing window informs sequencing of activity and sauna exposure.[5,7]
Clinical relevance. Because many xenobiotics are lipophilic—including POPs, certain legacy and modern pesticides, selected solvents, and some pharmaceuticals—they partition into adipose. Increased lipolysis can co-mobilize these lipophilic xenobiotics into circulation. The protocol pairs that window with elimination routes (dermal via sweat; biliary/enteric with bile-dependent binding) while minimizing enterohepatic recirculation through binder timing and adequate hydration/electrolytes.[6,8,9–16]
4. Protocol Application & Parameters
Core sequence. (1) Immediate-release niacin (titrated; see “Target Niacin Dose”), (2) light-to-moderate aerobic activity aligned to the rebound window, (3) sauna exposure with progressive heat conditioning, (4) strategic hydration/electrolytes, (5) micronutrient support, (6) binder timing to reduce enterohepatic recirculation. Adjustments include “flush-only” days for sensitive cases and staged ramp-ups for special populations.[6,8,17,18]
Protocol specifications (practitioner-directed)
- Target Niacin Dose (TND): practice-based guidance commonly references ~10 mg/kg body mass, with an empirical threshold around 500 mg (often sufficient in many women). Dosing is titrated to response and tolerance; receptor desensitization/tolerance is considered during multi-day courses.[6]
- Oil blend (composition & timing): flax (~60%), extra-virgin olive (~25%), walnut (~15%) by volume; taken after niacin and before exercise/sauna; doses scaled to body mass to support bile flow, emulsification, and digestive tolerance.[6,17]
- Lecithin: 2400 mg typical; may increase to ~3600 mg if signs of poor fat digestion. Taken between niacin and exercise to aid emulsification and support membranes; monitor GI tolerance.[17]
- Evening Primrose Oil (optional adjunct): individualized 500–1300 mg; timing ~2–2.5 h post-niacin alongside oils/lecithin. Rationale via GLA→DGLA→PGE1; include as an adjunct when clinically indicated.[17]
- Electrolytes & hydration: replace losses after each sauna cycle; include a Cal–Mag beverage (often during the second FIR cycle) and adequate sodium/potassium. Encourage measured intake based on baseline vitals, sweat rate, and weight change.[18]
- Binders: time to coincide with biliary flow and to minimize enterohepatic recirculation; selection varies by xenobiotic class and tolerance.[8,17]
Dermal route: The niacin flush produces cutaneous vasodilation that increases dermal perfusion, supporting sweat-based elimination during sauna; see human sweat-excretion evidence.[9–16]
5. Human Outcomes with Hubbard-Method Programs (POPs shown as exemplars of lipophilic xenobiotics)
Published human studies often emphasize persistent organic pollutants because they are analytically tractable (stable, bioaccumulative, with validated assays). This should be understood as an exemplar class: the physiologic sequencing targets lipophilic xenobiotics more broadly. For non-persistent chemicals, detecting biomarker change requires tight sampling windows relative to niacin/sauna timing and sometimes alternative matrices (e.g., sweat or bile-bound stool) in addition to blood.
| Year | Title & Authors | Population / Context | Design | Key Outcomes | Limitations | Source |
|---|---|---|---|---|---|---|
| 2007 | POPs exemplar Persistent organic pollutants in 9/11 World Trade Center rescue workers: reduction following detoxification (Dahlgren et al.) | n=7 WTC rescue workers; POPs profiled | Pre/post pilot (no control) | Several PCB congeners decreased at completion; mixed dioxin/furan shifts; symptom reporting improved. | Very small n; no control; limited follow-up; multiple comparisons. | [1] |
| 2012 | Occupational Methamphetamine exposure & chronic illness in police officers: improvement with sauna-based detoxification therapy (Ross & Sternquist) | n=69 officers with meth-lab exposures | Retrospective chart series (pre/post instruments) | Significant gains in SF-36, symptom severity, neurotoxicity scores; 92.8% completion; well-tolerated. | No randomization/control; self-report bias; heterogeneous exposures. | [2] |
| 2018 | Safety Safety & tolerability of sauna detoxification for protracted withdrawal (Lennox et al.) | Residential rehab cohort (HIV/HCV included) | Observational safety series | 99% completion; no heat illness; significant SF-36 physical/mental score gains at discharge. | Rehab setting confounds; no control; outcomes at discharge only. | [3] |
| 2019 | Pilot RCT A detoxification intervention for Gulf War Illness (Kerr, Carpenter et al.) | n=32 veterans randomized to intervention vs waitlist | Waitlist-controlled pilot RCT | Improved VR-36 physical (+≈6.9 points vs waitlist), multiple QoL/pain/fatigue scales; safe; benefits to 3 months in completers. | Small sample; waitlist control; feasibility-oriented power. | [4] |
6. Safety, Contraindications & Governance
Safety elements include inclusion/exclusion criteria; medication reconciliation; monitoring (vitals, hydration status, heat tolerance); escalation/de-escalation rules; adverse-event documentation; and practitioner training scope. Standard operating procedures: measure baseline vitals; track pre/post-sauna body weight to guide fluid replacement; assess orthostatic tolerance; and document adjustments. Available safety series and the pilot RCT reported high completion and no serious heat-related events, but extrapolation requires caution and appropriate clinical oversight.[3,4,18,17]
7. Comparative Approaches & Confounders
Distinctives include niacin-timed sequencing to the rebound-lipolysis window and pairing with dermal and enteric elimination. Potential confounders of both biomarkers and symptoms include concurrent weight loss, fasting, beta-agonist use, menstrual phase, baseline autonomic tone, and ambient heat exposure.[6,8,9–16]
8. Limitations & Research Gaps
Most studies to date are small and/or uncontrolled. Future priorities: adequately powered RCTs with active controls; standardized exposure characterization; harmonized outcome sets (symptoms, function, neurocognitive metrics); and targeted biomonitoring that discriminates metals from lipophiles and addresses enterohepatic cycling directly.[1,2,3,4]
Endnotes
- Dahlgren J, Cecchini M, Takhar H, Paepke O. Persistent organic pollutants in 9/11 World Trade Center rescue workers: reduction following detoxification. Chemosphere. 2007;69(8):1320–1325. doi:10.1016/j.chemosphere.2006.05.127. URL: https://pubmed.ncbi.nlm.nih.gov/17234251/
- Ross GH, Sternquist MC. Methamphetamine exposure and chronic illness in police officers: significant improvement with sauna-based detoxification therapy. Toxicol Ind Health. 2012;28(8):758–768. doi:10.1177/0748233711425070. Open access: https://pmc.ncbi.nlm.nih.gov/articles/PMC3573677/
- Lennox RD, et al. Safety and tolerability of sauna detoxification for the protracted withdrawal symptoms of substance abuse. Int J Gen Med. 2018;11:115–126. doi:10.2147/IJGM.S164486. Open access: https://pmc.ncbi.nlm.nih.gov/articles/PMC6259397/
- Kerr K, Morse G, Carpenter DO, et al. A detoxification intervention for Gulf War Illness: a pilot randomized controlled trial. Int J Environ Res Public Health. 2019;16(23):4884. doi:10.3390/ijerph16234884. Open access: https://pmc.ncbi.nlm.nih.gov/articles/PMC6862571/
- Wang W., et al. Effects of nicotinic acid on fatty acid kinetics, fuel selection, and pathways of glucose production in women. URL: https://www.ncbi.nlm.nih.gov/pubmed/10893322
- Pereira JN. The plasma free fatty acid rebound induced by nicotinic acid. Journal of Lipid Research. 1967;8(3):239–244. URL: http://www.jlr.org/content/8/3/239.full.pdf
- Tunaru S, Kero J, Schaub A, Wufka C, Blaukat A, Pfeffer K, Offermanns S. PUMA-G and HM74 are receptors for nicotinic acid and mediate its anti-lipolytic effect. Nature Medicine. 2003;9(3):352–355. URL: https://europepmc.org/article/med/12563315
- Cohn WJ, Boylan JJ, Blanke RV, et al. Treatment of chlordecone (Kepone) toxicity with cholestyramine. New England Journal of Medicine. 1978;298(5):243–248. URL: https://pubmed.ncbi.nlm.nih.gov/74014/
- Genuis SJ, Beesoon S, Birkholz D, et al. Biomonitoring and elimination of perfluorinated compounds and polychlorinated biphenyls through perspiration: Blood, urine, and sweat (BUS) study. ISRN Toxicology. 2013;2013:483832. doi:10.1155/2013/483832. URL: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3776372/
- Sears ME, Kerr KJ, Bray RI. Arsenic, cadmium, lead, and mercury in sweat: a systematic review. Journal of Environmental and Public Health. 2012;2012:184745. doi:10.1155/2012/184745. URL: https://www.hindawi.com/journals/jeph/2012/184745/
- Genuis SJ, Beesoon S, Birkholz D. Human excretion of bisphenol A: Blood, urine, and sweat (BUS) study. Journal of Environmental and Public Health. 2012;2012: Article ID. PMID:22253637. URL: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3255175/
- Genuis SJ, Beesoon S, Lobo RA, et al. Human excretion of polybrominated diphenyl ether flame retardants: Blood, urine, and sweat (BUS) study. BioMed Research International. 2017;2017:3676089. doi:10.1155/2017/3676089. URL: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5360950/
- Genuis SJ, Beesoon S, Birkholz D. Human elimination of phthalate compounds: Blood, urine, and sweat (BUS) study. The Scientific World Journal. 2012;2012:615068. doi:10.1100/2012/615068. URL: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3504417/
- Genuis SJ, et al. Human elimination of organochlorine pesticides: Blood, urine, and sweat (BUS) study. Journal of Environmental and Public Health. 2016;2016:1624643. doi:10.1155/2016/1624643. URL: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5069380/
- Zlateva S, Marinov P, Sabeva Y. Determination of toxic substances in sweat secret of severe forms of poisoning—Toxic coma. Journal of IMAB. 2007;13(1):81–83. URL (PDF): https://www.journal-imab-bg.org/statii-07/vol07_1_81-83str.pdf
- Genuis SJ. Elimination of persistent toxicants from the human body. Human & Experimental Toxicology. 2011;30(1):3–18. doi:10.1177/0960327110368417. URL (PDF): https://journals.sagepub.com/doi/pdf/10.1177/0960327110368417
- Detoxination® Practitioner Protocol Specifications & Special Circumstances. Internal curriculum document; proprietary and unpublished. Covers electrolyte and Cal–Mag timing, oil-blend composition and dosing, lecithin and optional EPO guidance, binder selection/timing, and case-specific adjustments. Access available to enrolled practitioners via the credentialed training program.
- Detoxination® Clinical Safety & Monitoring SOPs. Internal curriculum document; proprietary and unpublished. Details vitals monitoring, orthostatic assessment, heat-tolerance evaluation, adverse-event documentation, and weight-based fluid-replacement procedures. Access available to enrolled practitioners via the credentialed training program.
Daniel L. Root is the son of David E. Root, MD, MPH, the world-renowned Occupational Medicine specialist whose pioneering work in detoxification using niacin, exercise, and sauna formed the foundation of modern toxin-elimination science. Together the Roots refined and co-developed Detoxination®, the only clinically proven method shown to reduce accumulated drug residues, heavy metals, hazardous chemicals, radiation, and persistent organic pollutants.
Daniel authored Sauna Detoxification Using Niacin: Following The Recommended Protocol Of Dr. David E. Root and is CEO of Sabre Hawk, LLC. He trains practitioners in Detoxination and also serves as Assoc. Dir. of the AngioInstitute and Assoc. Editor of DETOXSCAN.org News, helping advance national efforts in exposure medicine and image-guided detoxification.